Interested in learning more about the possibilities of hydrogen education? Discover how the Horizon Hydrogen Grand Prix (H2GP) program can benefit students in a classroom near you. The result is for every 1 unit of hydrogen produced, 8 units of oxygen are created. Why does a hydrogen generator (electrolyzer) actually produce 8 times more oxygen than hydrogen? The molar mass of 2 molecules of water (2 H 20) is 18, and once the hydrogen generator splits the water into hydrogen and oxygen, the hydrogen (having a low atomic mass) only has a mass of 2 (H 2), while the oxygen has a mass of 16 (O 2). Students can understand, for instance, the process of hydrogen generation (electrolysis) with greater insight. Knowing the molar mass of hydrogen will open up a world of opportunity in terms of hydrogen education. Because of this apparent inertness, the H 2 molecule can be considered a very stable molecule at room temperature. Visualize trends, 3D orbitals, isotopes, and mix compounds. Interactive periodic table showing names, electrons, and oxidation states. Molecular hydrogen (H 2) can also react with many elements and compounds, but at room temperature, the reaction takes place at such a slow rate it's almost negligible. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. This is how hydrogen molecules – H 2 – are formed. Because of this reactivity, atoms of atomic hydrogen (H) combine with each other to form molecules of two atoms (H 2). This means that overall hydrogen is an extremely ‘light’ element. The more protons an atom has in its nucleus, the higher the atomic number, and the element also becomes heavier. As hydrogen gas is formed of two hydrogen (H) molecules bound together (H 2) – the molar mass can be calculated as 1.00784 x 2 = 2.01568 grams.Īs hydrogen consists of only one proton, its atomic number is 1. It can be calculated by multiplying the atomic weight of hydrogen (1.00784 atomic mass units) by 2. The molar mass of hydrogen is 2.01568 g/mole. This means you can simply multiply this by 4 moles, and you’ll find out how much carbon you’ll need to weigh. You already know carbon has a molar mass of 12.01 g/mol. Take this example: say you need 4 moles of pure carbon for an experiment. This means whether you’re trying to figure out how much carbon, titanium, or cobalt you need for an experiment – understanding the molar mass is essential. DO we seem to concentrate on the 'gram' as our go to guy on the periodic table for atomic weights and ultimately for molar masses and molecular weights Well the key here is the way we historically defined the mole.
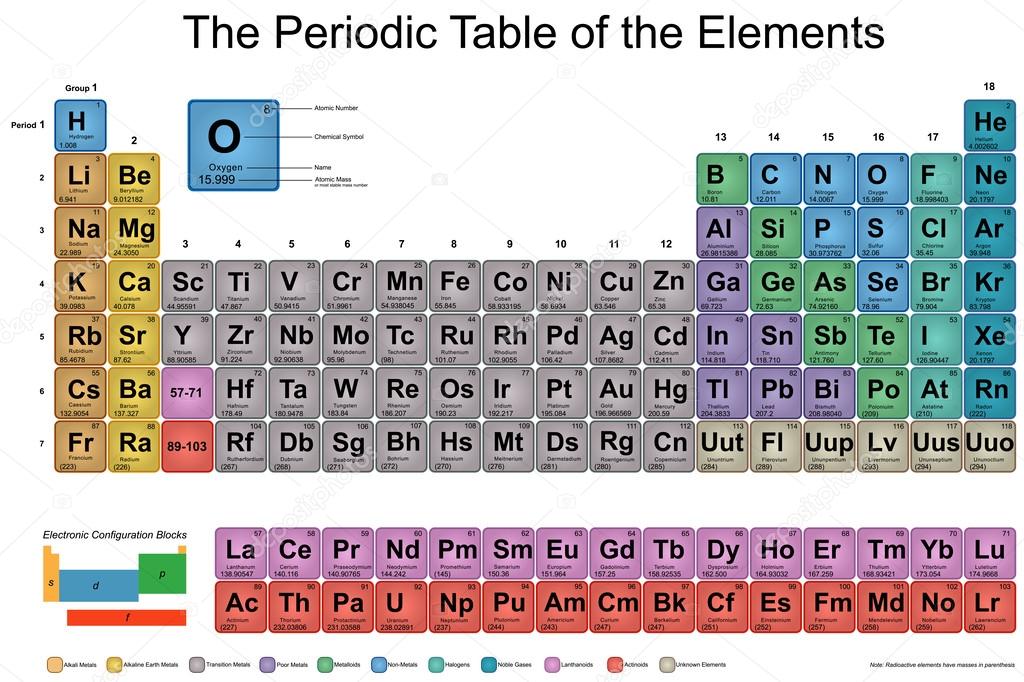


Molar Mass is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of a compound. The periodic table can often be presented with an abundance of data about each and every element listed.
